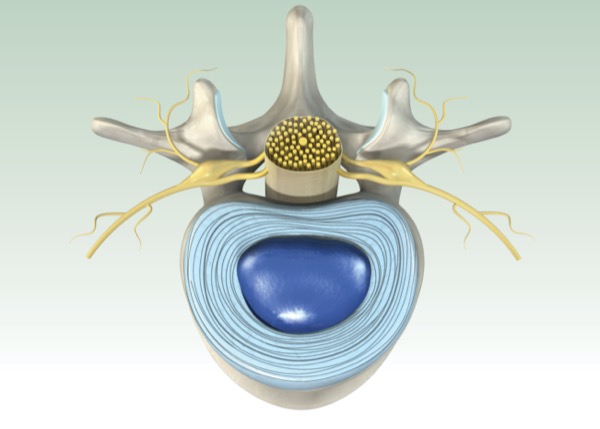
Degeneration of intervertebral discs (IVDs) is painful as cells of the inner disc transform to a subtype that is susceptible to or promotes infiltration of neurons from surrounding tissue, according to new in vitro and animal model investigations.
“We’ve identified for the first time particular cells that could be the key to understanding disc pain,” Dmitriy Sheyn, PhD, an assistant professor at the Board of Governors Regenerative Medicine Institute at Cedars-Sinai Medical