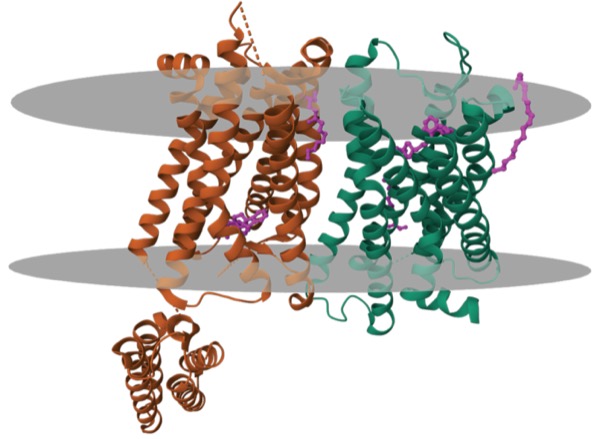
LAS VEGAS—A novel, first-in-class analgesic candidate in development for relief of acute pain could present less risk for abuse and addiction than current opioid-based agents, if promising results from a human addiction potential (HAP) trial,1 presented at the 2023 PAINWeek conference, are confirmed in evaluations that subject it to multiple potential modes of misuse.
“Based on all of our preclinical and human studies, we believe snorting or injecting cebranopadol would not make it