Originally published by our sister publication Specialty Pharmacy Continuum
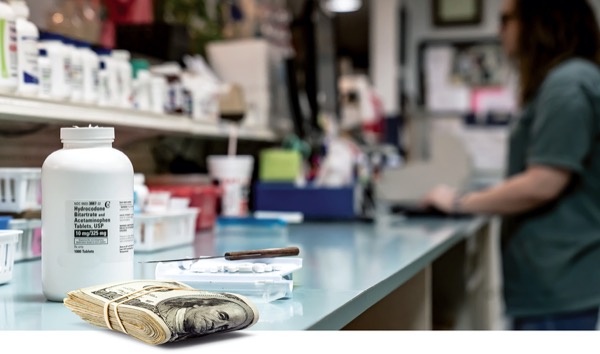
PHOENIX—Given enough cause, the Drug Enforcement Administration (DEA) will inspect pharmacies that fail to recognize and address suspicious prescriptions for opioids and other controlled substances (CS). Dispensers that ignore these “red flag” scripts subject their licenses, their pharmacies and themselves to DEA and state enforcement action, a CS compliance expert noted during