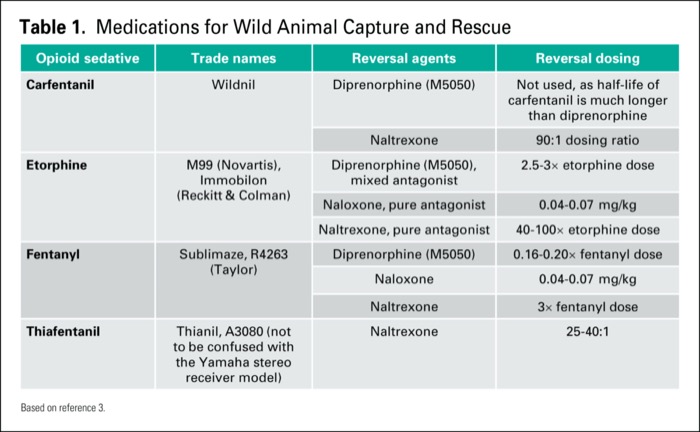
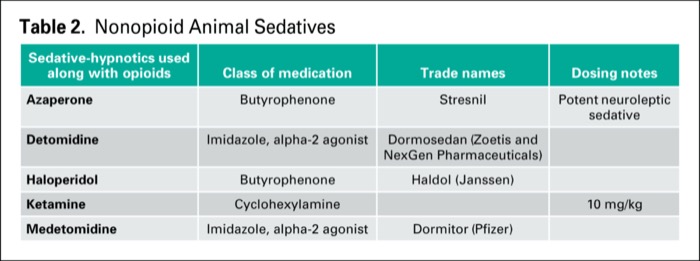
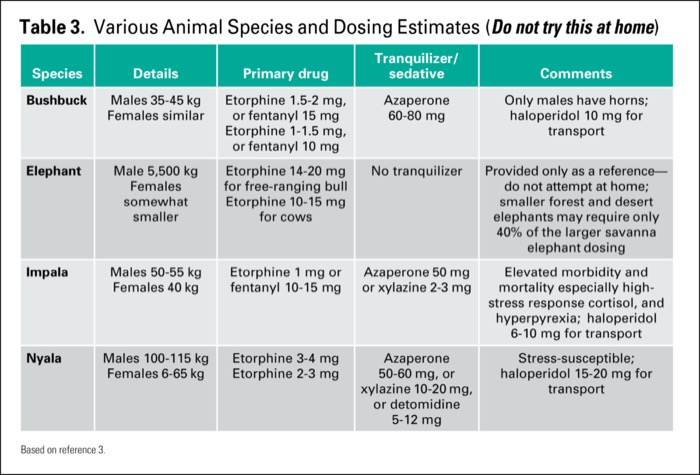
Originally published by our sister publication Anesthesiology News
Newport News, Va.
Have you ever wondered how wild animals like zebras or elephants are anesthetized? Well, I had a crash course in doing so.
So how does an American physician, an anesthesiologist, get involved in giving anesthesia to some of the wild animals of the African savanna? Not exactly a well-trod professional career path, but it came about as a result of doing some