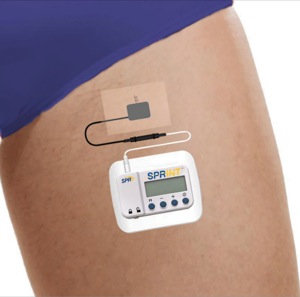
The FDA has cleared the SPRINT Peripheral Nerve Stimulation (PNS) System, maker SPR Therapeutics announced.
SPRINT is a minimally invasive, nonopioid, PNS system designed to provide relief for acute and chronic pain, according to SPR Therapeutics. The system consists of a threadlike lead and a matchbox-sized wearable stimulator. The lead is placed percutaneously with a fine needle, and electrical stimulation is delivered through the lead to activate the peripheral nerves