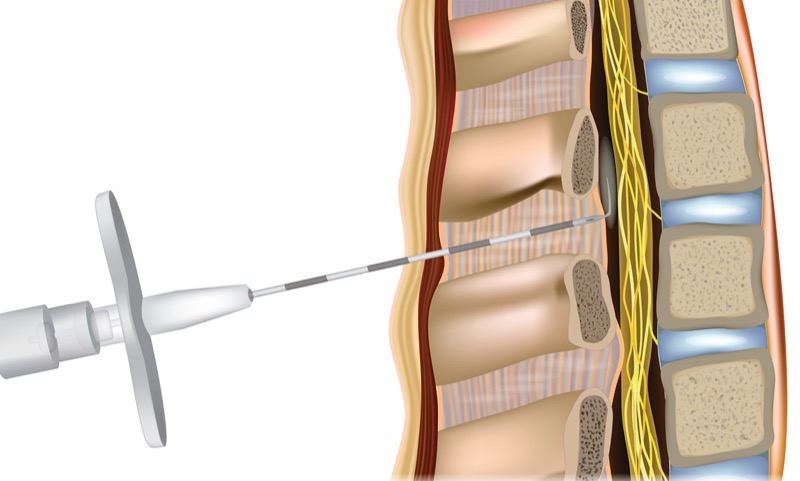
PHILADELPHIA—Despite advances in anesthesia care, the incidence of post-dural puncture headache (PDPH) remains troublingly high, affecting up to 40% of patients. Even though numerous reviews have addressed the prevention and management of PDPH, the condition is characterized by a lack of comprehensive recommendations.
To address this shortcoming, a multidisciplinary panel of 21 collaborators recently published consensus recommendations for the prevention, identification and management of