Originally published by our sister publication Specialty Pharmacy Continuum
The Institute for Safe Medication Practices (ISMP) took its safety message on the road at the NASP 2024 Annual Meeting & Expo, in Nashville, Tenn., where they presented several core practices that specialty pharmacists should follow to protect patients from drug harms.
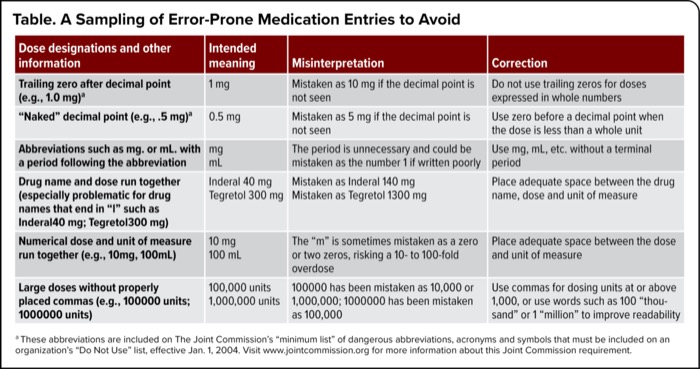
Jennifer Young, PharmD, BCPS, CSP, a medication safety specialist with ISMP, recommended that specialty pharmacies start by making use of