Denver—New research from Harvard University and Massachusetts General Hospital has concluded that the combination of opioids and medications with sedating side effects may negatively affect the efficacy of spinal cord stimulation.
Indeed, patients on higher doses of benzodiazepines and morphine equivalent dosing had less pain relief than their counterparts on lower doses of these agents.
As Charles A. Odonkor, MD, an interventional pain medicine fellow at Massachusetts General Hospital, in Boston, discussed at the 2019 annual meeting of the American Academy of Pain Medicine (AAPM), clinical guidelines currently recommend that patients taking opioids should be tapered off these agents before receiving spinal cord stimulation (SCS) therapy.
“Yet, adherence to these guidelines in clinical practice is highly variable,” Dr. Odonkor commented. “In practice, real-life data shows that patients receiving spinal cord stimulation therapy continue to remain on high doses of opioids, and in some cases, are concurrently using other sedating medications, including muscle relaxants. Beyond the potential hazards of this unfortunate practice, the impact on outcomes of spinal cord stimulation therapy remains unknown.”
Given such questions, Dr. Odonkor and his colleagues examined the impact of the concurrent use of opioids and sedating medications, such as benzodiazepines and muscle relaxants, on SCS outcomes.
In doing so, they collected data on 165 patients undergoing SCS trials. The participants were classified into two groups based on whether they used sedating medications (benzodiazepines or muscle relaxants) and whether their morphine equivalent dose was over 90 mg per day.
Reporting at the AAPM meeting (abstract LB003), Dr. Odonkor noted that the overall SCS trial success rate was 82%, with a median duration of seven days (range, three to 14 days).
It was found that patients on sedating medications were concomitantly on higher morphine equivalent doses (52.4 mg per day) than those who were not (24.6 mg per day; P=0.0004). These individuals also had lower percent mean pain relief (55% vs. 64%; P=0.048) with SCS trials.
“In other words, benzodiazepines and sedating medications increase the propensity for high levels of opioid consumption,” Dr. Odonkor said.
Conversely, patients who were taking fewer than 90 mg per day had greater mean pain relief than their counterparts on 90 mg or higher (62% vs. 49.9%; P=0.08).
Finally, the rate of SCS trial failure was greater among patients on sedating medications (20%) than among those who were not taking those agents (16%; P=0.054).
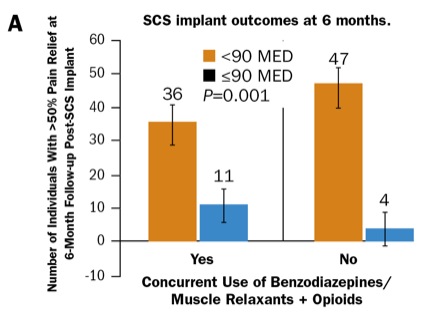
As Dr. Odonkor explained, the study also revealed that concomitant use of sedating medications and opioids decreased the success rates of SCS trials (13% vs. 87% for those two medications vs. single drugs; P=0.004) (Figure).
“Aside from the increased risk of morbidity, our research shows that concurrent use of opioids with benzodiazepines and/or muscle relaxants may lower the success rate of spinal cord stimulation therapy,” Dr. Odonkor told Pain Medicine News. “Interestingly, even among individuals using both opioids with benzodiazepines and/or muscle relaxants, the success rate of spinal cord stimulation therapy was higher among those on a lower morphine equivalent dose.”
According to Dr. Odonkor and senior author Shihab Ahmed, MD, the trial’s findings add new insight to the current literature by highlighting that less recognized muscle relaxants may have a codependent relationship with benzodiazepines, similar to what is known about codependent use of opioids and benzodiazepines.
“While it is previously known that smoking diminishes analgesic response to spinal cord stimulation therapy, the confounding factors for this effect remain elusive,” he said. “We found during subgroup analysis that spinal cord stimulation failure rates were higher among patients with dual use of opioids and benzodiazepines/opioids who were also smokers. This shows a potential cross-interaction between smoking, opioid use and sedating medications.
“We suspect the high failure rates in this cohort of patients may be due to underlying hyperactivation of inflammatory stress, which interferes with response to spinal cord stimulation therapy,” he added. “This remains an area of active research, and future large randomized studies may help paint a clearer picture regarding the underlying mechanisms of the observed effects.”
Given these findings, the investigators recommended that their peers who provide SCS therapy be cognizant of the risks of concurrent use of these medications. In instances where combined use of these medications is unavoidable, they noted that a lower dose of opioids with sedating antispasmodics should be considered.
“This may help reduce risks and decrease spinal cord stimulation failure rates,” Dr. Odonkor noted. “In addition, given the codependence of muscle relaxants/benzodiazepines with opioid use, clinicians should endeavor to taper patients off these medications prior to initiating spinal cord stimulation therapy.”
For David Provenzano, MD, the president of Pain Diagnostics and Interventional Care in Bridgeville, Pa., the study helps demonstrate that risk factors for interventional care may be broader than many clinicians think. “What I take away from this study is that while there are common risk factors for interventional care, we also have to look at patients’ medication usage and see if we can modify those factors prior to intervening, because they can negatively affect outcomes,” Dr. Provenzano said.
As he discussed, drug-weaning strategies may one day prove useful in SCS. “We don’t really know yet if weaning strategies are successful for surgical outcomes,” he said. “But we do know, at least from retrospective studies in the joint replacement population, that preoperative drug weaning is associated with better outcomes.
“In the end, pain specialists can’t simply jump into this thinking they can use an interventional device to help patients get off their medications,” Dr. Provenzano added. “I think you need to have a preoperative plan prior to considering some of these devices. And you have to look at the whole patient and address medical issues, comorbidities and medication usage, because it clearly affects outcomes.”
—Michael Vlessides
Dr. Odonkor reported no relevant financial disclosures. Dr. Provenzano has served as a consultant to Biotronik, Boston Scientific, Medtronic and Nevro, and has received research support from Abbott, Medtronic and Nevro.